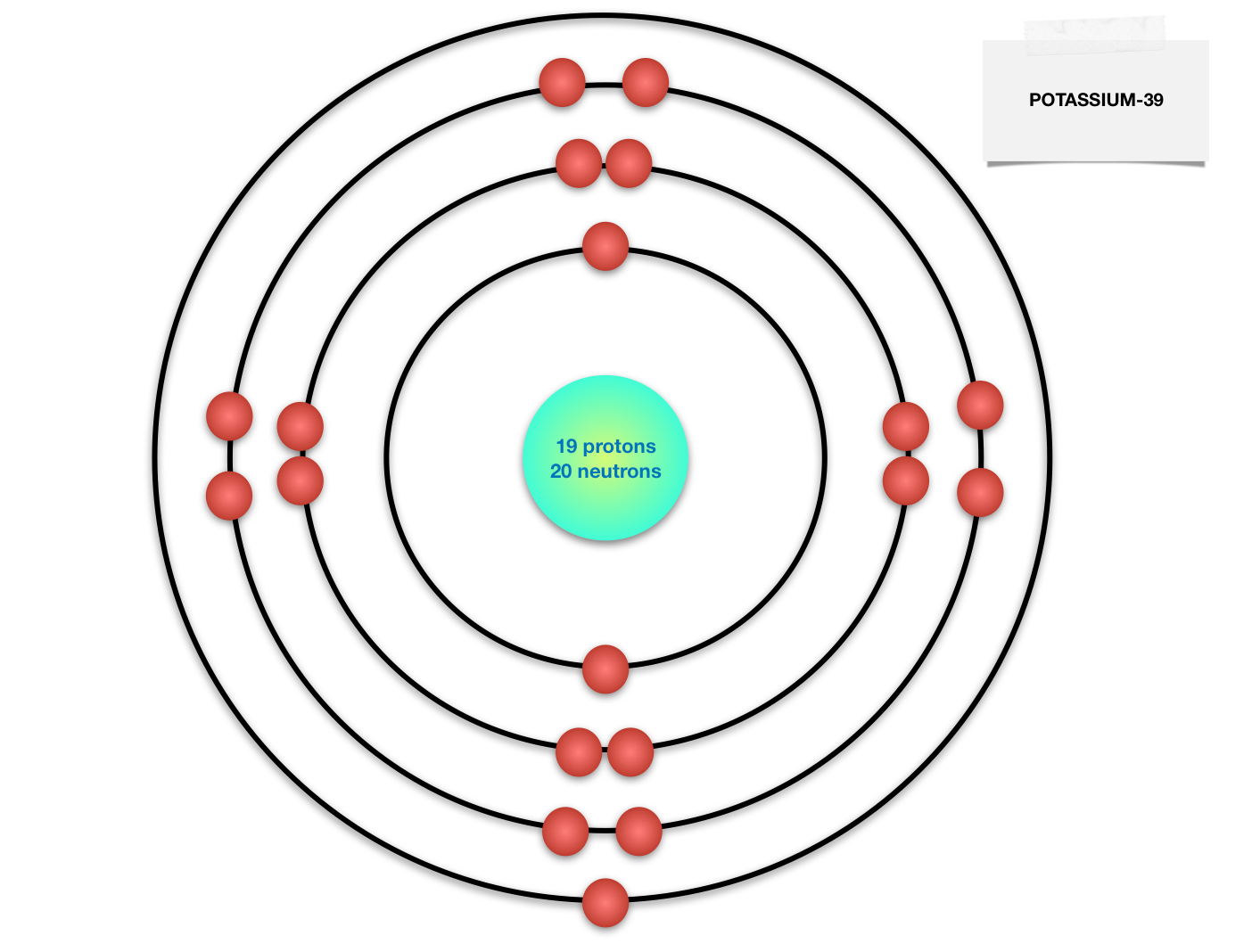
The Bohr model of the atom, a radical departure from earlier, classical descriptions, was the first that incorporated quantum theory and was the predecessor of wholly quantum-mechanical models. The protons and neutrons are placed inside the nucleus while the electrons are housed in three shells i.e. It comes to rest in the \(n = 6\) orbit, so \(n_2 = 6\). Historically, Bohr’s model of the hydrogen atom is the very first model of atomic structure that correctly explained the radiation spectra of atomic hydrogen. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr. Conclusion The final Bohr model of the Sulfur atom consists of 16 protons, 16 neutrons, and 16 electrons. 
In this case, the electron starts out with \(n = 4\), so \(n_1 = 4\). Bohrs model calculated the following energies for an electron in the shell, n. What is the energy (in joules) and the wavelength (in meters) of the line in the spectrum of hydrogen that represents the movement of an electron from Bohr orbit with n = 4 to the orbit with n = 6? In what part of the electromagnetic spectrum do we find this radiation? This is the final Bohr model of fluorine atom as we have used all 9 electrons: 2 electrons in the 1 st electron shell and 7 electrons in the 2 nd electron. The Bohr Model has an atom consisting of a small, positively charged nucleus orbited by negatively charged electrons. Encircling this nucleus are two electron shells, carrying a total of 9 electrons. Repeat for p, d, and f.\): Calculating Electron Transitions in a One–electron System In the fluorine Bohr model, the nucleus holds 9 protons and 10 neutrons. Simply make a column for all the s orbitals with each n shell on a separate row. 
When Bohr calculated his theoretical value for the Rydberg constant, R, R, and compared it with the experimentally accepted value, he got excellent agreement. Figure 2.3.5 The Emission Spectra of Elements Compared with Hydrogen These images show (a) hydrogen gas, which is atomized to hydrogen atoms in the discharge tube (b) neon and (c) mercury. which is identical to the Rydberg equation in which R k h c. Bohr Atom sim activity.doc - 43 kB Download all files as a compressed. This chart is straightforward to construct. Scientists needed a fundamental change in their way of thinking about the electronic structure of atoms to advance beyond the Bohr model. 1 1 H Hydrogen 1.01 2 3 4 Li Be Lithium Beryllium 6.94 9.01 11 12 Na Mg Sodium Magnesium 22.99 24.31 19 20 K Ca Potassium Calcium 39.10 40.08 37 38 Rb Sr Rubidium Strontium. 
\) The arrow leads through each subshell in the appropriate filling order for electron configurations. Book 2: Atoms, The Periodic Table & Bohr Models 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
